Following the Centers for Medicare & Medicaid Services’ (CMS) announcement of draft guidance on its highly anticipated reimbursement decision regarding Aduhelm (aducanumab) and other future anti-amyloid monoclonal antibody (mAb) products in Alzheimer’s disease (AD); Leia Mais »
International News
Duchenne muscular dystrophy drug pipeline variation is promising, says GlobalData
By 2030, patients living with Duchenne muscular dystrophy (DMD) — a genetic muscle wasting disease caused by mutations on the gene that produces dystrophin — will have greater choice when it comes to the drug types used for treatment, according to GlobalData. The leading data and analytics company notes that there are 23 DMD-targeting therapeutics currently being developed in the US and Japan, and eight of these have the potential to make it to market in the next 10 years. Leia Mais »
Datwyler makes its debut at fce pharma with high-quality, system-critical components for vaccine providers
Middletown, DE, December 1, 2021 – At FCE Pharma (November 30 – December 2, 2021; São Paulo) Datwyler, (Booth G098) will highlight its high-quality, system-critical elastomer components for injectable drugs such as the COVID-19 vaccine. Since the start of the pandemic, Datwyler has supplied system-critical components including stoppers and aluminum seals to vaccine developers around the world. Leia Mais »
Chinese pharma companies eye strategic alliances to bring innovative siRNA-based therapeutics, observes GlobalData
The overall potential of siRNA-based therapeutics has attracted Chinese pharma companies to tap opportunities and bring promising innovative therapies across the globe. Against this backdrop, the Chinese pharma companies have increased their focus towards the siRNA pipeline through strategic alliances*, finds GlobalData, a leading data and analytics company. Leia Mais »
Eosinophilic esophagitis market is evolving rapidly in the US, says GlobalData
There are currently no Food and Drug Administration (FDA) approved treatments for eosinophilic esophagitis (EoE) in the US, says GlobalData. The leading data and analytics company notes that pipeline drugs with diverse mechanisms of action (MOAs) in late-stage development will create a dynamic market with a variety of treatment options for EoE patients in the near future. Leia Mais »
Busch China celebrates 20th anniversary
( Maulburg, )
Shanghai was then and still is today one of the largest industrial centers in China. The international airport and the port of Shanghai are important trading hubs for Busch. The steady growth and ever-increasing popularity of the vacuum pump manufacturer were the main reasons for moving into Busch’s new headquarters in China in 2017. The Zizhu National High-tech Industrial Development Zone has been home to the Chinese headquarters ever since. In addition to office space, Busch Shanghai has a large and well-equipped workshop for the inspection, maintenance and testing of vacuum pumps, compressors, and blowers of all types. All after-sales services can be carried out quickly and reliably on site. A dedicated System Building Department not only takes care of the construction and design of vacuum systems, but also the installation and commissioning at the customer’s site. This ensures that individual customer requirements are adapted to the strict Busch standards.
During the sudden outbreak of COVID-19 in January last year, Busch Shanghai rushed twelve MINK claw vacuum pumps to the hastily constructed hospital -Wuhan Huoshenshan Hospital. These pumps were used to supply medical vacuum for Corona patients.
In addition to the medical sector, products from Busch Vacuum Solutions are used in almost all industrial sectors, such as chemicals, semiconductors, medical technology, plastics and the food industry. The Busch Group is a family-owned company whose management remains entirely in the hands of the Busch family to this day. More than 3,500 employees work for Busch Vacuum Solutions in over 60 companies and more than 40 countries worldwide.
Busch is proud to be able to celebrate the 20th anniversary of the Sales Company in China with an internal event for the employees. It is very important for Busch to be present in the Chinese market with its own products with the best possible quality. Being close to the local customers is just one of the many success factors for Busch in China.
Tate & Lyle PLC half year results for the 2022 financial year
Strategic transformation progressing well; strong growth in Food & Beverage Solutions
Exclusive interview with Professor Sir John Bell announced as Life Science Integrates celebrates ten years of Pharma Integrates
London, UK, 3rd November 2021 / Sciad Newswire / Life Science Integrates (LSI) is delighted to announce the exciting agenda for their flagship event, Pharma Integrates, held live online on 16th November 2021. Leia Mais »
Merck & Co leads global R&D race for use of immune checkpoint inhibitors after surgery, says GlobalData
Immune checkpoint inhibitors (ICIs) have transformed the treatment of many late-stage cancers. This success has caused pharma companies to invest heavily into investigating their use in an earlier setting after a tumor has been removed by surgery (adjuvant), says GlobalData. The leading data and analytics company notes that, among these companies, Merck & Co has taken the most proactive steps for its mega blockbuster drug, Keytruda, to ensure dominance in the adjuvant setting.
Sakis Paliouras, PhD, Senior Oncology Analyst at GlobalData, comments: “So far, in most patients who receive any adjuvant treatment after surgery, that is going to be chemotherapy, radiation, or a combination of both. ICIs are widely anticipated to receive marketing authorizations in numerous adjuvant settings, and in many cancers, a first-mover advantage could bring billions in extra revenue.”
Currently, the highest-grossing ICI, Merck & Co’s Keytruda, which brought in $14.4bn in global revenue for 2020, has gained an FDA label in three adjuvant settings: melanoma, bladder cancer, and triple-negative breast cancer. Bristol Myers-Squibb’s (BMS) Opdivo also has three FDA labels in the adjuvant setting, while the nearest competitor, Roche’s Tecentriq, has gained only one label so far, in adjuvant treatment of non-small cell lung cancer (NSCLC), based on results from the IMpower010 trial.
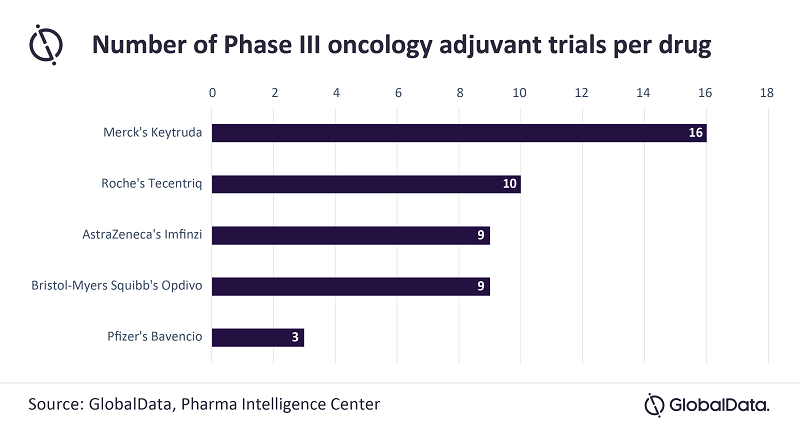
Paliouras continues: “An analysis of late-stage clinical trials in the adjuvant setting shows that Keytruda has the upper hand, with 16 ongoing clinical trials in various cancers, followed by Tecentriq with ten trials and Imfinzi/Opdivo with nine. However, AstraZeneca has taken a more proactive approach by sponsoring more trials than Roche, which has relied more on academic collaboration.
“Adjuvant treatment can be very lucrative and if any of these trials prove an extension in overall survival of patients, high physician adoption is to be expected. The area with the most adjuvant trials is cancers of the gastrointestinal tract, which is where the next big battle of ICIs is expected to be fought. Including liver, gastrointestinal, and colorectal cancer among others, this segment will include hundreds of thousands of patients worldwide eligible to be treated with ICIs.”
Growth potential for India in innovation and R&D, says GlobalData
India has the third largest pharma market in the world by volume, according to research by GlobalData, placing it behind giants such as the US and China. The leading data and analytics company notes that India’s potent manufacturing capabilities, high export activity and large generics market have supported the country in grabbing ‘bronze place’. Leia Mais »
 2A+ Farma Portal de notícias
2A+ Farma Portal de notícias
